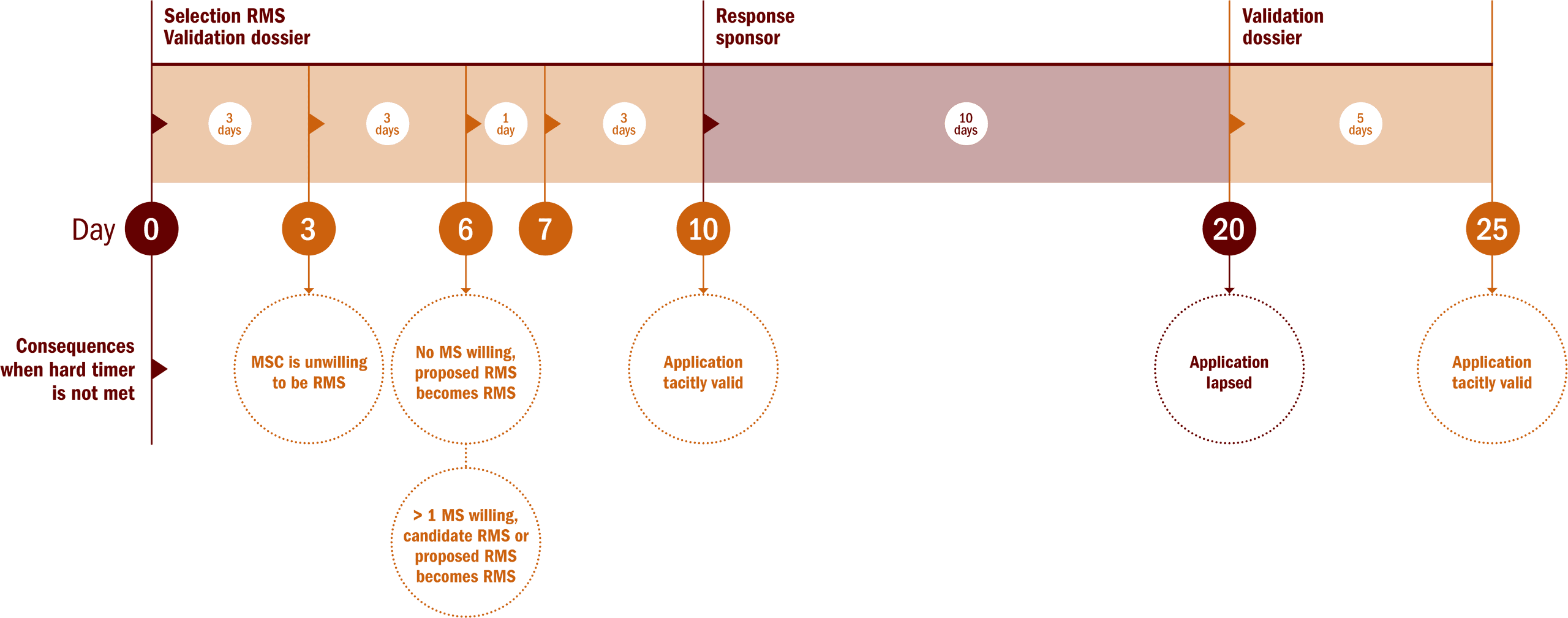
The maximum period for the validation phase is 25 days including 15 days for the MS (10 plus an additional 5 in case of the rMS requests for information), and 10 days for the sponsor to complete the application dossier.
During the validation phase, the rMS is selected for multinational clinical trials. If a MS does not meet its time limits there will be a tacit approval i.e. the clinical trials falls within the scope and the application dossier is complete. If a sponsor does not meet its time limits the application automatically lapses.
Overview of timelines
The procedure of selection of a rMS in a multinational clinical trial is not fully presented here. See for complete procedure CTIS training module 6 Selection of reporting Member State (RMS) and validation of the clinical trial application.