Clinical investigation into the safety and/or (clinical) performance of the medical device falls within the scope of the EU Medical Device Regulation (MDR). Other research in which medical devices are used as part of a study (for example, to measure an endpoint), but in which the safety, performance and/or effectiveness of the medical device itself are not investigated, falls outside the scope of the MDR. These studies may fall under the scope of the Dutch Medical Research Involving Human Subjects Act [Wet Medisch-wetenschappelijk onderzoek met mensen, WMO] or the Dutch Embryo Act [Embryowet].
Definition clinical investigation
The MDR gives the following definition of clinical investigation:
Systematic investigation involving one or more human subjects, undertaken to assess the safety or performance of a medical device.
Frames for clinical investigation
The MDR has the following three frames for clinical investigation, with different requirements and procedures:
- Clinical investigations carried out for conformity purposes (i.e. in the context of product development and obtaining/extending a CE marking) (MDR article 62 and article 74.2)
- Post-Market Clinical Follow-up (PMCF) study involving invasive and/or burdensome procedures, also referred to as PMCF investigations (MDR article 74.1)
- Other clinical investigations with medical devices that are not conducted in the context of conformity purposes or PMCF (MDR article 82; supplemented by national rules from the WMO)
To determine the framework that applies to your clinical investigation, you may use the flowchart below. An explanatory note is available.
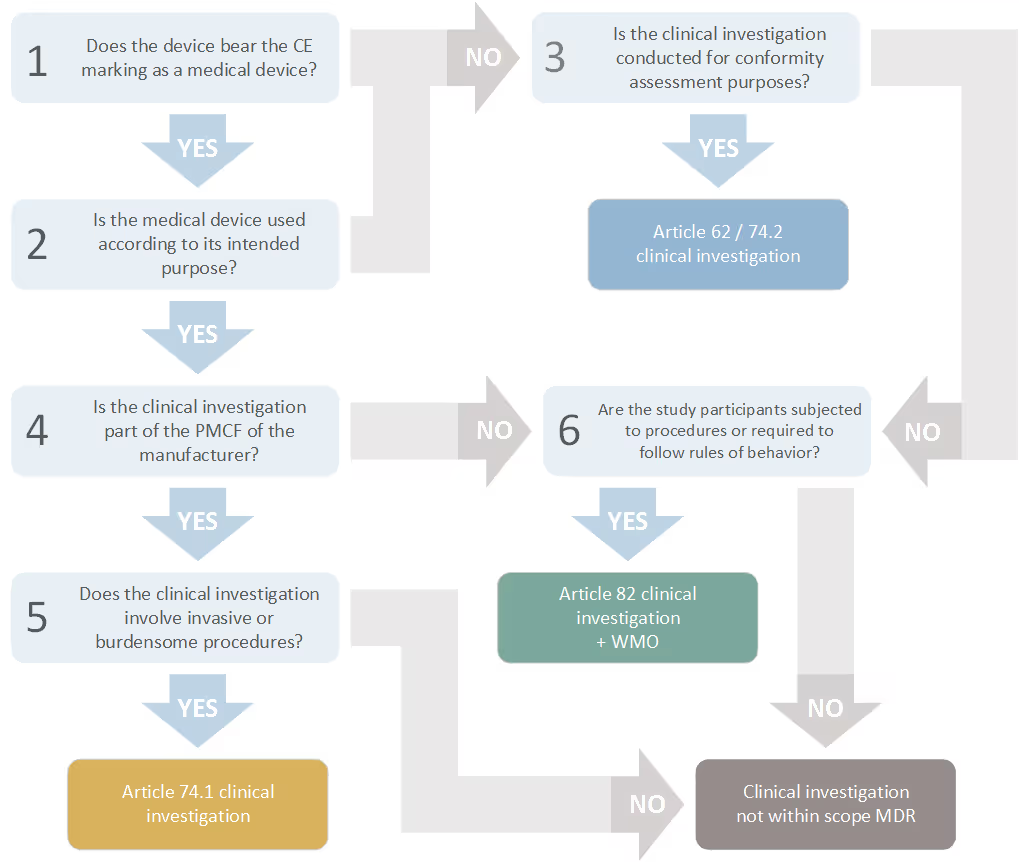
Image: © CCMO